Asuris Esteem (PPO) Asuris TruAdvantage + Rx Primary (PPO) Asuris TruAdvantage + Rx Classic (PPO) PPO Plan Provider Directory
MUSEO FIELD DE HISTORIA NATURAL CHICAGO, ILLINOIS EEUU y MUSEO CONTISUYO MOQUEGUA, PERU INFORME DE CAMPO E INFORME FINAL PROYECT

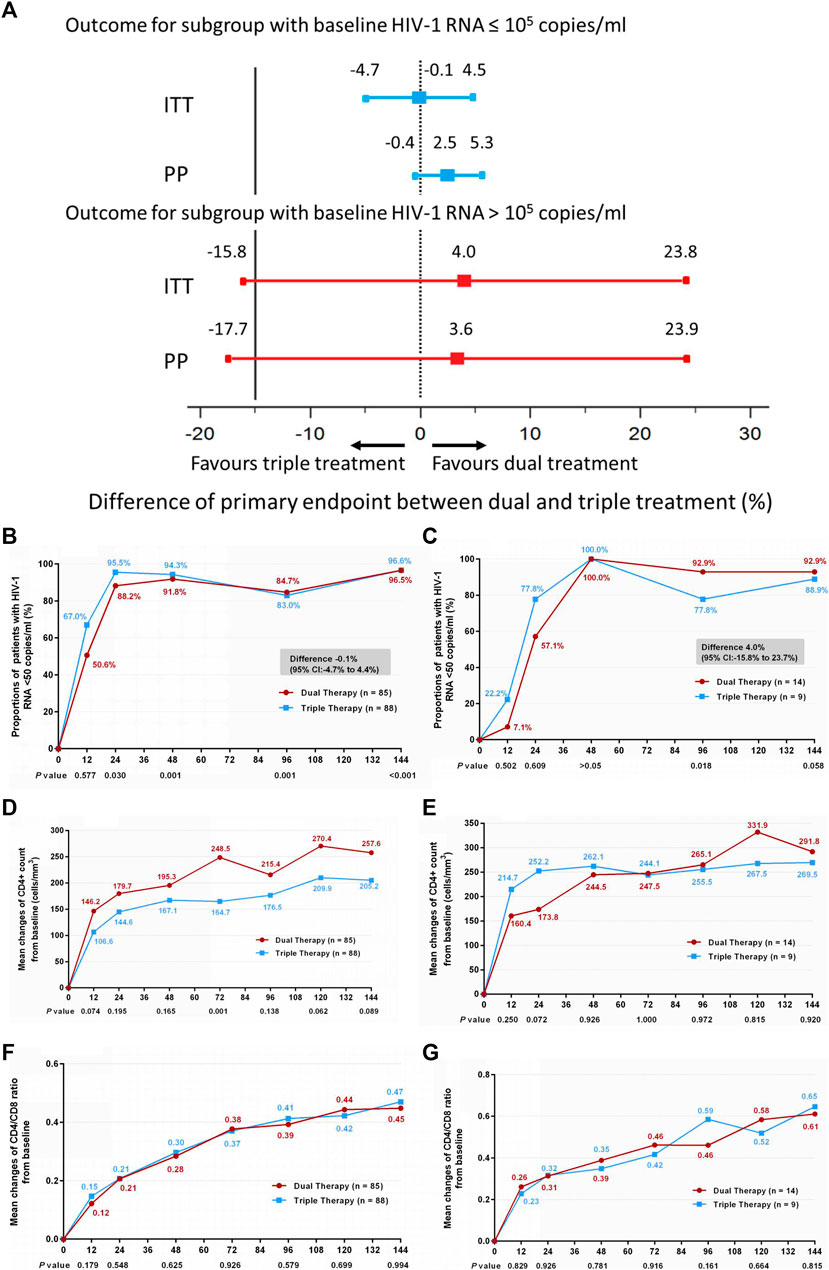
Frontiers | Antiretroviral Long-Term Efficacy and Resistance of Lopinavir/Ritonavir Plus Lamivudine in HIV-1-Infected Treatment-Naïve Patients (ALTERLL): 144-Week Results of a Randomized, Open-Label, Non-Inferiority Study From Guangdong, China